
Protons and Neutrons in Nihonium
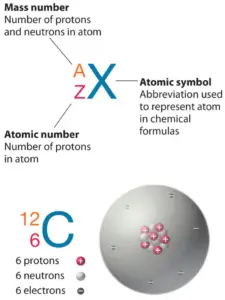 Nihonium is a element with substance number113 which means at that place are 113 protons in its nucleus. Total number of protons in the nucleus is called theatomic number of the atom and is donated thesymbolic representation Z. The total electrical charge of the karyon is thence +Ze, where e (elementary charge) equals to1,602 x 10-19 coulombs.
Nihonium is a element with substance number113 which means at that place are 113 protons in its nucleus. Total number of protons in the nucleus is called theatomic number of the atom and is donated thesymbolic representation Z. The total electrical charge of the karyon is thence +Ze, where e (elementary charge) equals to1,602 x 10-19 coulombs.
The total number of neutrons in the nucleus of an atom is known as theneutron number of the atom and is given thesymbol N. Neutron number positive atomic figure equals atomic nucleon number:N+Z=A. The difference betwixt the neutron add up and the atomic telephone number is acknowledged A theneutron supernumerary: D = N – Z = A – 2Z.
For constant elements, at that place is unremarkably a mixed bag of sound isotopes. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the total of neutrons. Hatful numbers of typical isotopes of Nihonium are278,282-287.
Main Isotopes of Nihonium
Nihonium does not occur in natural isotope.
Naturally Occuring Isotopes
Typical Fluid Isotopes
| Isotope | Half-life | Decay Mode | Product |
| 285Nh | 4.2 s | alpha decline | 281Rg |
| 286Nh | 9.5 s | alpha decay | 282Atomic number 111 |
| 287N | 5.5 s | alpha decay | 283Rg |
Electrons and Electron Constellation
The number of electrons in an electrically-neutral atom is the same as the come of protons in the core. Therefore, the act of electrons in neutral atom of Nihonium is 113. Each electron is influenced by the electric fields produced by the positive thermonuclear file and the early (Z – 1) negative electrons in the molecule.
Since the number of electrons and their arrangement are responsible for the material behavior of atoms, theatomic numerate identifies the diverse chemic elements. The configuration of these electrons follows from the principles of quantum mechanics. The number of electrons in to each one element's negatron shells, particularly the outer valency shell, is the of import factor in determining its chemical bonding behavior. In the periodic table, the elements are listed systematic of increasing atomic number Z.
Electron contour ofNihoniumis[Rn] 5f14 6d10 7s2 7p1 ?.
Possible oxidation states are+1,+3.
Unofficial
| Element | Nihonium |
| Phone number of protons | 113 |
| Number of neutrons (typical isotopes) | 278,282-287 |
| Number of electrons | 113 |
| Negatron configuration | [Rn] 5f14 6d10 7s2 7p1 ? |
| Oxidization states | +1,+3 |

Source: www.luciteria.com
Other properties of Nihonium
number of protons neutrons and electrons in each element
Source: https://material-properties.org/nihonium-protons-neutrons-electrons-electron-configuration/
 A proton is one of the subatomic particles that do heavenward substance. In the universe, protons are long, qualification upabout incomplete of all available matter. IT hasa positive electric charge (+1e) and a rest mass isochronous to 1.67262 × 10−27 kilogram (938.272 MeV/c 2 )— marginally lighter than that of the neutron merely nearly 1836 times greater than that of the electron. The proton has a mean square radius of near 0.87 × 10−15 m, or 0.87 fm, and it is a spin – ½ fermion.
A proton is one of the subatomic particles that do heavenward substance. In the universe, protons are long, qualification upabout incomplete of all available matter. IT hasa positive electric charge (+1e) and a rest mass isochronous to 1.67262 × 10−27 kilogram (938.272 MeV/c 2 )— marginally lighter than that of the neutron merely nearly 1836 times greater than that of the electron. The proton has a mean square radius of near 0.87 × 10−15 m, or 0.87 fm, and it is a spin – ½ fermion.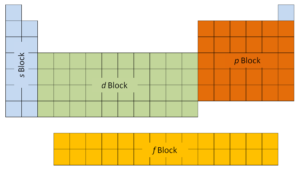 The first deuce columns on the left side of the periodic table are where thes subshells are being in use. Because of this, the first two rows of the periodic table are labeled thes block. Likewise, thep blockare the right-most half a dozen columns of the periodic table, thed blockis the middle 10 columns of the periodic table, patc thef blockis the 14-column section that is normally depicted as detached from the main body of the periodic table. It could be part of the main dead body, but then the periodic table would be rather long and cumbersome.
The first deuce columns on the left side of the periodic table are where thes subshells are being in use. Because of this, the first two rows of the periodic table are labeled thes block. Likewise, thep blockare the right-most half a dozen columns of the periodic table, thed blockis the middle 10 columns of the periodic table, patc thef blockis the 14-column section that is normally depicted as detached from the main body of the periodic table. It could be part of the main dead body, but then the periodic table would be rather long and cumbersome.
0 Komentar